Electrolysis of water is slowed down by weightlessness, says S.Ananthanarayanan.
Creating oxygen by passing electricity is an expensive source, but when in outer space, it could a good way to help astronauts breathe, as well as burn fuel. And it would be the only method to support an outpost located on the Moon, or on Mars.
How fast oxygen gets generated, however, is found to depend on the force of gravity. As the gravity on the Moon or on Mars is much less than it is on the Earth, it could be a problem to sustain an establishment on these planets. Bethany A. Lomax, Gunter H. Just, Patrick J. McHugh, Paul K. Broadley, Gregory C. Hutchings, Paul A. Burke, Matthew J. Roy, Katharine L. Smith and Mark D. Symes, from the University of Glasgow, European Space Research Centre, Noordwijk, Nederlands, University of Manchester and John Hopkins University in Maryland, USA, describe in the journal, Nature Communications, the nature of this problem and how it is being studied. The subject has become important, the paper says, in the light of increasing interest in creating a presence on the Moon, and evidence that water may be a local resource.
The oxygen atom, when by itself, has a tendency to ‘become whole’, either pairing with another oxygen atom, to form O2, or by combining with a complementary atom like hydrogen, carbon or iron, to form oxides, like water (H2O), CO2, or the oxide of iron, which is rust. And the oxide form is the low energy, stable form. And hence, to prise the oxygen atom out of the oxide, it takes energy. A well-known instance, where oxygen separates from CO2 is photosynthesis, where green plants pull the oxygen out of the CO2 molecule with the help of energy supplied by sunlight. And another method is electrolysis, where oxygen is separated from H2O with the energy supplied by passing an electric current.
The nature of the water molecule, which is like other oxides, is that the atom of hydrogen has given up an electron, which has a negative charge, and the oxygen atom has gained this electron. The atom of hydrogen is hence left positively charged, while the oxygen atom has a negative charge. And some of the molecules split, with the positively charged hydrogen atoms and the negatively charged oxygen atoms floating free. Now, if there are two plates in the water, with a difference of voltage, the charged atoms are drawn to the positive and negative ends, where the charges are neutralised, and the gases are set free.
So where does gravity enter the picture? Well, when the oxygen atoms reach the positive end, they give up the negative charge they are carrying and then pair up, as oxygen gas. But, as oxygen gas, they are not attracted to one side or the other, and they would ‘stay put’, with molecules of oxygen collecting as bubbles of gas. The effect of a layer of gas around the plate with the positive voltage would then be to prevent more negatively charged oxygen atoms from reaching the plate and the formation of the gas would stop.
Fortunately, even if electrical forces do not move oxygen gas away, the pressure of the water does push the gas bubble upwards, in the same way as the ‘up thrust’ on any object that is immersed in a liquid. And the reason for this pressure, of the liquid, is nothing but gravity, which is pulling the liquid down, which acts to fill the space occupied by the bubble.
The bubbles of oxygen then stream upwards, where they can be captured and bottled. And the bubble moving away clears the positive plate, which can go on with the good work of taking up the negative charge from oxygen atoms of water molecules, and set them free, as gas. We can imagine that the stronger the force of gravity, pressing down on the water, the faster the production of oxygen gas.
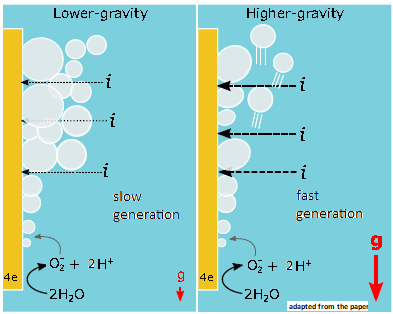
But in outer space, far from massive objects like the Earth, gravity is feeble and electrolysis would stop as soon as it starts. And on the Moon, or on Mars, where the gravity is just one sixth and one third of the gravity on the earth, respectively, electrolysis would be slow. And given this fact, the scientists who design the facilities at these outposts would need to know how much electrolysis gets retarded when gravity drops, to half, or a tenth, or, specifically, to one third or one sixth of the gravity on the Earth.
Now, how can experiments be carried out to test electrolysis under reduced gravity? One way to conduct the experiment is in a laboratory that is in free fall. Inside such a chamber, the so-called drop tower, there is no gravity, or very low gravity. But the free fall down a tower that is, say 100 m high, would last just 2.5 seconds. The paper speaks of the more common method of creating weightlessness in an aircraft in a ‘parabolic flight’, where the aircraft moves forward, but is in free fall. In this method, the drops can be considerably more than 100 m, and last up to 22 seconds. And experiments have been conducted on electrolysis under very low gravity. The trouble, the paper says, is that it is not clear that these very low gravity results can be extended to work out what it would be like in the gravity on the Moon or on Mars. And while better data is possible in parabolic flights, the experiments are expensive, cumbersome and short-lived.
Trials involving higher gravity, however, are easier to organise – using a centrifuge, or a rapidly rotating container. The centrifugal force experienced amounts to increased gravity, pointing away from the centre of rotation, and this can be maintained for prolonged periods. High gravity, or hyper gravity, experiments were hence conducted to see how electrolysis behaved between low gravity levels and gravity eight times as strong as on the Earth. And the findings at different levels of gravity were compared.
The results, the paper says, show that there is a uniform trend of the efficiency of oxygen generation rising as the level of gravity increases. Although this can be expected, it is not evident without being verified, the paper says. Based on the experiments, however, it was found that it is true and the more reliable data from ground-based centrifuges can be used to work out what it would be like at the gravity found on the Moon, the paper says.
And it is found that an electrolytic cell working on the Moon, where gravity is a sixth of what it is on the Earth, would generate 11% less oxygen. This is a modest reduction and it shows that the additional power that the cell would consume on the Moon is not substantial.
It looks like “appropriately designed ground-based hypergravity systems can be used to determine the ideal operating conditions for a given system in low gravity, potentially negating the need for costly and complex parabolic flight experiments,” the paper says.
------------------------------------------------------------------------------------------ Do respond to : response@simplescience.in-------------------------------------------